Terminology
/General
Viscosity vs Rheology: Understanding the Difference
Explore the concepts of viscosity and rheology, their key differences, and how they are measured. Learn about their applications in fluid and material analysis.
2 min read
Advertisement
Table of Contents
When we talk about fluids, whether it’s the water we drink or the air we breathe, we often simplify things in physics to make calculations easier. This leads us to the concept of an “ideal fluid.” But how does an ideal fluid compare to the real fluids we encounter every day? Let’s break it down.
An ideal fluid is a theoretical concept. We define it as a fluid that is both incompressible and non-viscous. This means a few key things:
It’s important to note: Ideal fluids are imaginary and don’t exist in nature. They are simply a useful simplification for theoretical calculations. They exhibit smooth, laminar flow, which is depicted below:
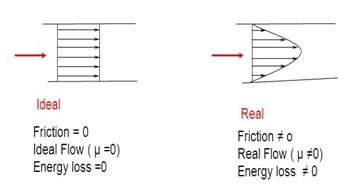
Now, let’s talk about fluids we actually see in the real world. Real fluids are, well, real. They possess properties that ideal fluids lack:
Examples of real fluids are everywhere: kerosene, petrol, and even castor oil.
Here’s a table to clearly outline the differences between ideal and real fluids:
| Characteristic | Ideal fluid | Real fluid |
|---|---|---|
| Compressible | No | Yes |
| Viscosity | No | Yes |
| Surface Tension | No | Yes |
| Flow Type | Laminar | Turbulent |
| Friction | No | Yes |
In essence, ideal fluids are a useful theoretical construct that helps simplify fluid dynamics calculations. However, it is important to remember that real fluids have characteristics like viscosity and compressibility, which affect their behavior.
Advertisement
Terminology
/General
Explore the concepts of viscosity and rheology, their key differences, and how they are measured. Learn about their applications in fluid and material analysis.
Terminology
/General
Explore the differences between dynamic and kinematic viscosity, their formulas, units, and relationship to fluid density and flow.